In-situ Raman spectroscopy applied for the cyclohexane oxidation
The selective oxidation of cyclohexane is an important process in chemical industry. The products cyclohexanol and cyclohexanone are necessary for the caprolactam synthesis, a monomer for the polyamide-6 production. The intermediate product during the cyclohexane oxidation, cyclohexylhydroperoxide, and cyclohexanol and cyclohexanone are easier oxidable than cyclohexane. Therefore, the reaction is carried out with a conversion lower than 6 % in order to get a selectivity of cyclohexanol and cyclohexanone of 70 – 90 %.
In spite of the technical importance, the detailed reaction mechanism is not yet fully clarified. Therefore, at the institute of micro process engineering laser Raman spectroscopy was applied to examine this reaction under defined conditions in a microchannel at 80 bars and up to 240 °C. The picture shows the experimental setup with a microscope (left picture) and the microreactor (right picture).
The microreactor consists of a silicon wafer, in which a meander-shaped rectangular microchannel (0.525 mm x 0.525 mm) was etched. Thereon, a Borofloat glass plate was anodically bonded. This construction allows a pressure up to 80 bars. Cyclohexane is driven by Helium at 80 bars and a mass-flow controller. Oxygen is dosed with a second mass-flow controller at 80 bars up to the limit of solubility. The microreactor can be heated by oil from the lower side up to 240 °C.
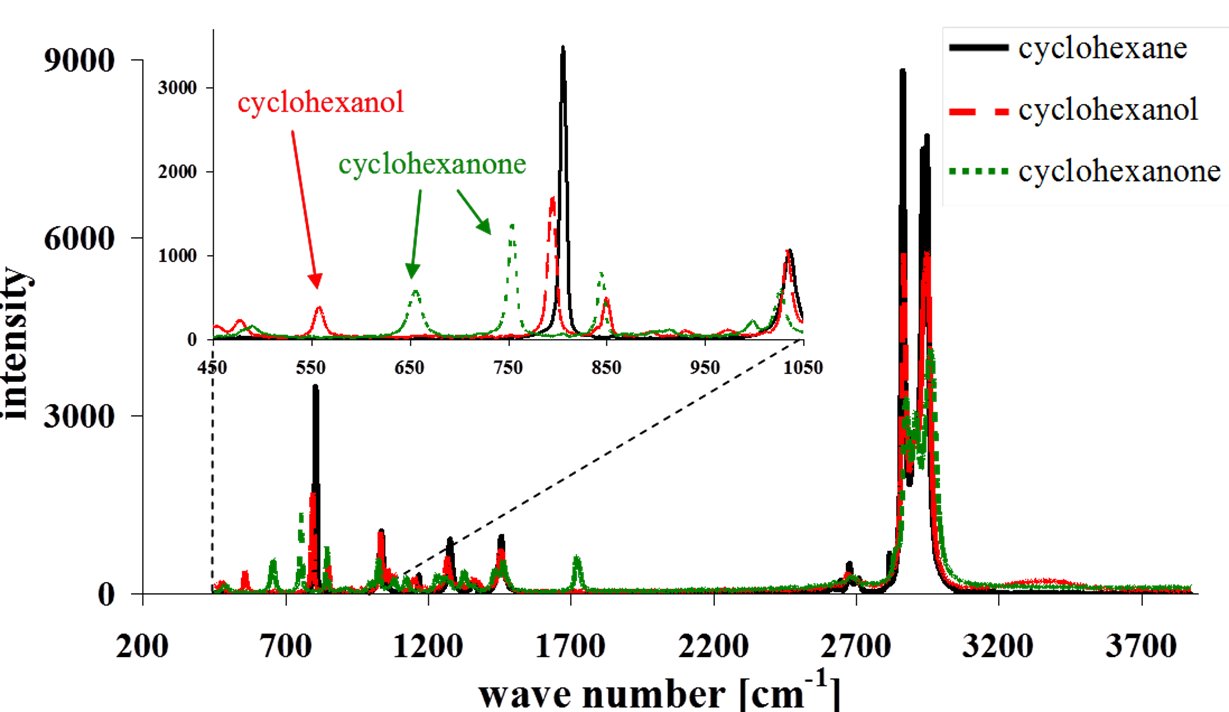
The radiation of a continuous-wave argon ion laser (488 nm, 100 mW, blue ray in picture) with small bandwidth is leaded through a dichroitic mirror and the microscope and focused into the microchannel. The molecules in the microchannel emit Raman stray light, which is leaded back via the microscope and the dichroitic mirror (red ray in picture) to a spectrometer with cooled CCD camera. The following picture shows a typical Raman spectrum, from which concentration profiles along the reaction channel can be determined.
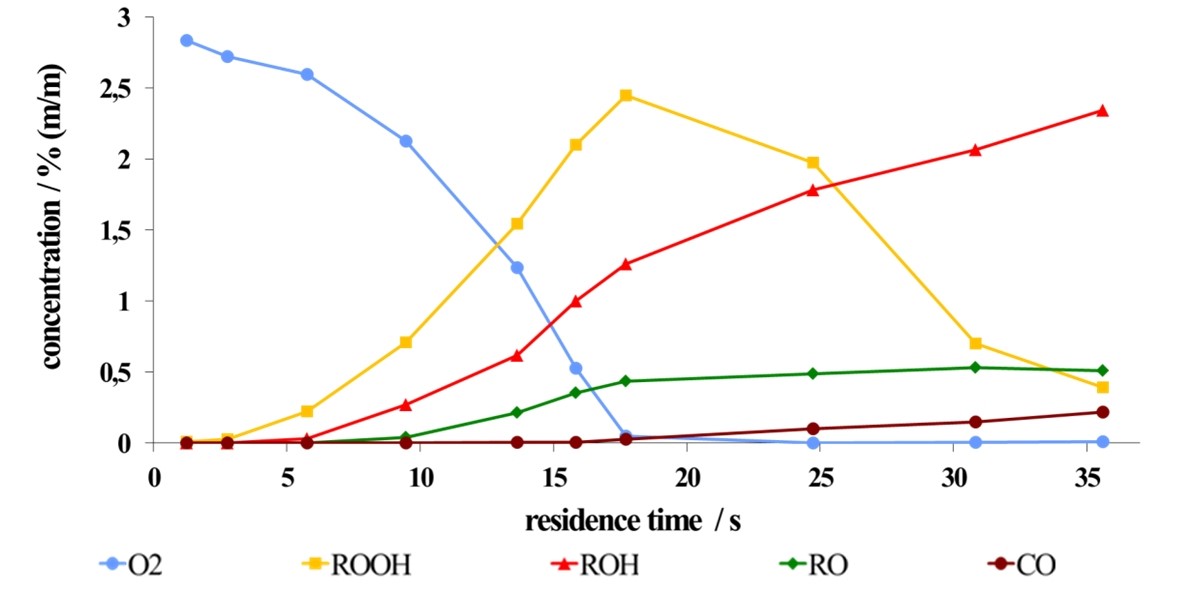
The next picture shows a concentration profile of the products cyclohexylhydroperoxide (ROOH), cyclohexanol (ROH), cyclohexanone (RO), carbonmonoxide (CO) and the educt oxygene (O2) along the reaction channel as a function of the residence time.