Power-to-Methanol: Experimental and modeling work for CO2 capture and its application in combination with methanol synthesis
- chair:
-
place:
Master's Thesis/student assistant
-
institute:
IMVT
-
starting date:
as of now
- Kontaktperson:
-
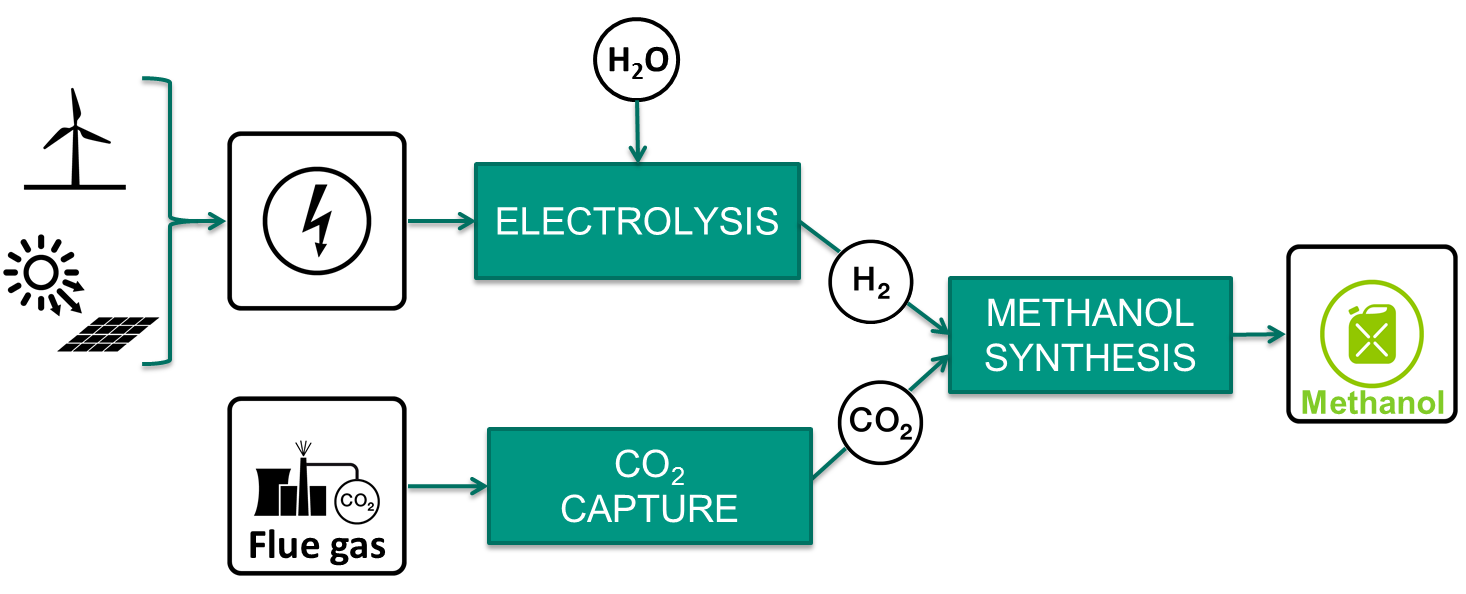
Figure 1: Scheme of the Power-to-Methanol concept using CO2 from flue gas, renewable electricity and water as feedstock.
Our unsustainable energy system and its effect on climate change is forcing our society to transform the energy system to sustainable during the 21st century. The main contributor to the climate change is the emission of greenhouse gases, mainly CO2, into the atmosphere. Reduction of CO2 emissions by decreasing the use of fossil fuel can be done by increasing the share of renewable energy and electrification of transportation and industry. However, electrification of industries with energy intensive processes, such as iron and cement industries, is difficult to accomplish and their emissions represent a high share of global emissions. Carbon capture and utilization (CCU) technologies capture the CO2 from emissions or the atmosphere and then, use it to manufacture fuels and chemicals. CCU for methanol production is one of the most relevant CCU applications owing this to the relevance of methanol as a basic chemical. With a global annual production of over 110 million tons, methanol is used for production of fuels, chemical intermediates, plastics and fine chemicals. Methanol can be directly produced from H2 and CO2 by the following reaction:
Equation 1: Methanol synthesis reaction equation.
Power-to-Methanol (PtM) technology (Figure 1) is a synergetic solution for both storing intermittent renewable energy such as wind or solar, and recycling CO2 captured from flue gases or air. This technology uses H2 produced by water electrolysis using renewable energy, and CO2 captured from industrial flue gases to produce methanol. In order for PtM to succeed, the production cost of methanol need to be reduced. This can be accomplished by simplifying the process and thus, reducing the equipment required and the energy consumed. For this reason, the Institute for Micro Process Engineering (IMVT) is developing a novel hybrid process which couples CO2 capture with methanol synthesis. This process significantly simplifies the PtM technology and thus, making it potentially economically feasible.
In this Master thesis, the capture of CO2 by a liquid physical solvent will be investigated both with experimental and modelling work. The experimental work will be performed in an experimental set up built at the IMVT. The modelling work will be executed in Aspen Plus® using an existing model as starting point. Experimental work combining the CO2 capture part with the methanol synthesis part will be performed if the time allows it. The student can choose to write the thesis either in German or English.
Requirements: Basic knowledge in laboratory work and Microsoft Office applications (Word, Excel and Power Point).
Useful: Previous experience with modeling and simulation.
Examiner: Prof. Dr.-Ing. Roland Dittmeyer
Supervisor: Dr. Francisco Vidal Vázquez
Contact: Francisco.vidal-vazquez@kit.edu
Processing date: 4th Apr 2022
